Xenobiotic metabolism
Xenobiotic metabolism (from the Greek xenos "stranger" and biotic "related to living beings") is the set of metabolic pathways that modify the chemical structure of xenobiotics, which are compounds foreign to an organism's normal biochemistry, such as drugs and poisons. These pathways are a form of biotransformation present in all major groups of organisms, and are considered to be of ancient origin. These reactions often act to detoxify poisonous compounds; however, in some cases, the intermediates in xenobiotic metabolism can themselves be the cause of toxic effects.
Xenobiotic metabolism is divided into three phases. In phase I, enzymes such as cytochrome P450 oxidases introduce reactive or polar groups into xenobiotics. These modified compounds are then conjugated to polar compounds in phase II reactions. These reactions are catalysed by transferase enzymes such as glutathione S-transferases. Finally, in phase III, the conjugated xenobiotics may be further processed, before being recognised by efflux transporters and pumped out of cells.
The reactions in these pathways are of particular interest in medicine as part of drug metabolism and as a factor contributing to multidrug resistance in infectious diseases and cancer chemotherapy. The actions of some drugs as substrates or inhibitors of enzymes involved in xenobiotic metabolism are a common reason for hazardous drug interactions. These pathways are also important in environmental science, with the xenobiotic metabolism of microorganisms determining whether a pollutant will be broken down during bioremediation, or persist in the environment. The enzymes of xenobiotic metabolism, particularly the glutathione S-transferases are also important in agriculture, since they may produce resistance to pesticides and herbicides.
Contents |
Permeability barriers and detoxification
That the exact compounds an organism is exposed to will be largely unpredictable, and may differ widely over time, is a major characteristic of xenobiotic toxic stress.[1] The major challenge faced by xenobiotic detoxification systems is that they must be able to remove the almost-limitless number of xenobiotic compounds from the complex mixture of chemicals involved in normal metabolism. The solution that has evolved to address this problem is an elegant combination of physical barriers and low-specificity enzymatic systems.
All organisms use cell membranes as hydrophobic permeability barriers to control access to their internal environment. Polar compounds cannot diffuse across these cell membranes, and the uptake of useful molecules is mediated through transport proteins that specifically select substrates from the extracellular mixture. This selective uptake means that most hydrophilic molecules cannot enter cells, since they are not recognised by any specific transporters.[2] In contrast, the diffusion of hydrophobic compounds across these barriers cannot be controlled, and organisms, therefore, cannot exclude lipid-soluble xenobiotics using membrane barriers.
However, the existence of a permeability barrier means that organisms were able to evolve detoxification systems that exploit the hydrophobicity common to membrane-permeable xenobiotics. These systems therefore solve the specificity problem by possessing such broad substrate specificities that they metabolise almost any non-polar compound.[1] Useful metabolites are excluded since they are polar, and in general contain one or more charged groups.
The detoxification of the reactive by-products of normal metabolism cannot be achieved by the systems outlined above, because these species are derived from normal cellular constituents and usually share their polar characteristics. However, since these compounds are few in number, specific enzymes can recognize and remove them. Examples of these specific detoxification systems are the glyoxalase system, which removes the reactive aldehyde methylglyoxal,[3] and the various antioxidant systems that eliminate reactive oxygen species.[4]
Phases of detoxification
The metabolism of xenobiotics is often divided into three phases: modification, conjugation, and excretion. These reactions act in concert to detoxify xenobiotics and remove them from cells.
Phase I - modification
In phase I, a variety of enzymes acts to introduce reactive and polar groups into their substrates. One of the most common modifications is hydroxylation catalysed by the cytochrome P-450-dependent mixed-function oxidase system. These enzyme complexes act to incorporate an atom of oxygen into nonactivated hydrocarbons, which can result in either the introduction of hydroxyl groups or N-, O- and S-dealkylation of substrates.[5] The reaction mechanism of the P-450 oxidases proceeds through the reduction of cytochrome-bound oxygen and the generation of a highly-reactive oxyferryl species, according to the following scheme:[6]
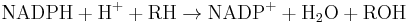
Phase II - conjugation
In subsequent phase II reactions, these activated xenobiotic metabolites are conjugated with charged species such as glutathione (GSH), sulfate, glycine, or glucuronic acid. These reactions are catalysed by a large group of broad-specificity transferases, which in combination can metabolise almost any hydrophobic compound that contains nucleophilic or electrophilic groups.[1] One of the most important of these groups are the glutathione S-transferases (GSTs). The addition of large anionic groups (such as GSH) detoxifies reactive electrophiles and produces more polar metabolites that cannot diffuse across membranes, and may, therefore, be actively transported.
Phase III - further modification and excretion
After phase II reactions, the xenobiotic conjugates may be further metabolised. A common example is the processing of glutathione conjugates to acetylcysteine (mercapturic acid) conjugates.[7] Here, the γ-glutamate and glycine residues in the glutathione molecule are removed by Gamma-glutamyl transpeptidase and dipeptidases. In the final step, the cystine residue in the conjugate is acetylated.
Conjugates and their metabolites can be excreted from cells in phase III of their metabolism, with the anionic groups acting as affinity tags for a variety of membrane transporters of the multidrug resistance protein (MRP) family.[8] These proteins are members of the family of ATP-binding cassette transporters and can catalyse the ATP-dependent transport of a huge variety of hydrophobic anions,[9] and thus act to remove phase II products to the extracellular medium, where they may be further metabolised or excreted.[10]
Endogenous toxins
The detoxification of endogenous reactive metabolites such as peroxides and reactive aldehydes often cannot be achieved by the system described above. This is the result of these species' being derived from normal cellular constituents and usually sharing their polar characteristics. However, since these compounds are few in number, it is possible for enzymatic systems to utilize specific molecular recognition to recognize and remove them. The similarity of these molecules to useful metabolites therefore means that different detoxification enzymes are usually required for the metabolism of each group of endogenous toxins. Examples of these specific detoxification systems are the glyoxalase system, which acts to dispose of the reactive aldehyde methylglyoxal, and the various antioxidant systems that remove reactive oxygen species.
History
Studies on how people transform the substances that they ingest began in the mid-nineteenth century, with chemists discovering that organic chemicals such as benzaldehyde could be oxidized and conjugated to amino acids in the human body.[11] During the remainder of the nineteenth century, several other basic detoxification reactions were discovered, such as methylation, acetylation, and sulfonation.
In the early twentieth century, work moved on to the investigation of the enzymes and pathways that were responsible for the production of these metabolites. This field became defined as a separate area of study with the publication by Richard Williams of the book Detoxication mechanisms in 1947.[12] This modern biochemical research resulted in the identification of glutathione S-transferases in 1961,[13] followed by the discovery of cytochrome P450s in 1962,[14] and the realization of their central role in xenobiotic metabolism in 1963.[15][16]
See also
- Drug design
- Drug metabolism
- Microbial biodegradation
- Biodegradation
- Bioremediation
- Antioxidant
- SPORCalc, an example process for exploring xenobiotic and drug metabolism databases[17]
References
- ^ a b c Jakoby WB, Ziegler DM (5 December 1990). "The enzymes of detoxication". J. Biol. Chem. 265 (34): 20715–8. PMID 2249981. http://www.jbc.org/cgi/reprint/265/34/20715.
- ^ Mizuno N, Niwa T, Yotsumoto Y, Sugiyama Y (2003). "Impact of drug transporter studies on drug discovery and development". Pharmacol. Rev. 55 (3): 425–61. doi:10.1124/pr.55.3.1. PMID 12869659. http://pharmrev.aspetjournals.org/cgi/content/full/55/3/425#C.%20Role%20of%20Transporters%20in%20Drug%20AbsorptionA.
- ^ Thornalley PJ (1 January 1990). "The glyoxalase system: new developments towards functional characterization of a metabolic pathway fundamental to biological life". Biochem. J. 269 (1): 1–11. PMC 1131522. PMID 2198020. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1131522.
- ^ Sies H (1997). "Oxidative stress: oxidants and antioxidants". Exp Physiol 82 (2): 291–5. PMID 9129943. http://ep.physoc.org/cgi/reprint/82/2/291.pdf.
- ^ Guengerich FP (2001). "Common and uncommon cytochrome P450 reactions related to metabolism and chemical toxicity". Chem. Res. Toxicol. 14 (6): 611–50. doi:10.1021/tx0002583. PMID 11409933.
- ^ Schlichting I, Berendzen J, Chu K, et al. (2000). "The catalytic pathway of cytochrome p450cam at atomic resolution". Science 287 (5458): 1615–22. doi:10.1126/science.287.5458.1615. PMID 10698731.
- ^ Boyland E, Chasseaud LF (1969). "The role of glutathione and glutathione S-transferases in mercapturic acid biosynthesis". Adv. Enzymol. Relat. Areas Mol. Biol. 32: 173–219. doi:10.1002/9780470122778.ch5. PMID 4892500.
- ^ Homolya L, Váradi A, Sarkadi B (2003). "Multidrug resistance-associated proteins: Export pumps for conjugates with glutathione, glucuronate or sulfate". Biofactors 17 (1-4): 103–14. doi:10.1002/biof.5520170111. PMID 12897433.
- ^ König J, Nies AT, Cui Y, Leier I, Keppler D (1999). "Conjugate export pumps of the multidrug resistance protein (MRP) family: localization, substrate specificity, and MRP2-mediated drug resistance". Biochim. Biophys. Acta 1461 (2): 377–94. doi:10.1016/S0005-2736(99)00169-8. PMID 10581368.
- ^ Commandeur JN, Stijntjes GJ, Vermeulen NP (1995). "Enzymes and transport systems involved in the formation and disposition of glutathione S-conjugates. Role in bioactivation and detoxication mechanisms of xenobiotics". Pharmacol. Rev. 47 (2): 271–330. PMID 7568330.
- ^ Murphy PJ (1 June 2001). "Xenobiotic metabolism: a look from the past to the future". Drug Metab. Dispos. 29 (6): 779–80. PMID 11353742. http://dmd.aspetjournals.org/cgi/content/full/29/6/779.
- ^ Richard Tecwyn Williams. 20 February 1909-29 December 1979 A. Neuberger, R. L. Smith Biographical Memoirs of Fellows of the Royal Society, Vol. 28, Nov., 1982 (Nov., 1982), pp. 685-717
- ^ Booth J, Boyland E, Sims P (1 June 1961). "An enzyme from rat liver catalysing conjugations with glutathione". Biochem. J. 79 (3): 516–24. PMC 1205680. PMID 16748905. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1205680.
- ^ Omura T, Sato R (1 April 1962). "A new cytochrome in liver microsomes". J. Biol. Chem. 237 (4): 1375–6. PMID 14482007. http://www.jbc.org/cgi/reprint/237/4/PC1375.
- ^ Estabrook RW (2003). "A passion for P450s (remembrances of the early history of research on cytochrome P450)". Drug Metab. Dispos. 31 (12): 1461–73. doi:10.1124/dmd.31.12.1461. PMID 14625342. http://dmd.aspetjournals.org/cgi/content/full/31/12/1461.
- ^ Estabrook, R. W., Cooper, D. Y., and Rosenthal, O. (1963). "The light reversible carbon monoxide inhibition of steroid C-21 hydroxylase system in adrenal cortex.". Biochem. Z. 338: 741–55. PMID 14087340.
- ^ James Smith; Viktor Stein (2009). "SPORCalc: A development of a database analysis that provides putative metabolic enzyme reactions for ligand-based drug design". Computational Biology and Chemistry 33 (2): 149–159. doi:10.1016/j.compbiolchem.2008.11.002. PMID 19157988.
Further reading
- H. Parvez and C. Reiss (2001). Molecular Responses to Xenobiotics. Elsevier. ISBN 0-345-42277-5.
- C. Ioannides (2001). Enzyme Systems That Metabolise Drugs and Other Xenobiotics. John Wiley and Sons. ISBN 0-471-89466-4.
- M. Richardson (1996). Environmental Xenobiotics. Taylor & Francis Ltd. ISBN 0-748-40399-X.
- C. Ioannides (1996). Cytochromes P450: Metabolic and Toxicological Aspects. CRC Press Inc. ISBN 0-849-39224-1.
- Y.C. Awasthi (2006). Toxicology of Glutathionine S-transferses. CRC Press Inc. ISBN 0-849-32983-3.
External links
Databases
- Drug metabolism database
- Directory of P450-containing Systems
- University of Minnesota Biocatalysis/Biodegradation Database
Drug metabolism
Microbial biodegradation
History